Glycan Release
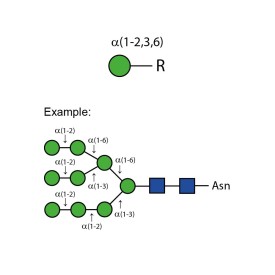
α-(1-2,3,6) mannosidase
E-AM01
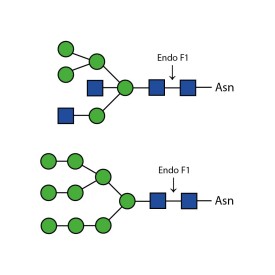
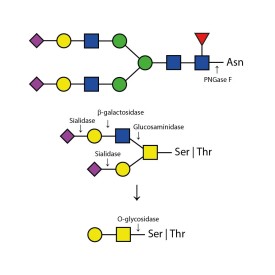
Endoglycosidase F1
E-EF01
LudgerLiberate hydrazinolysis kit
LL-HYDRAZ-A2
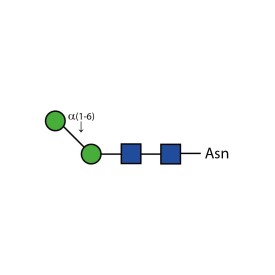
α-(1,6) core mannosidase
E-AM02
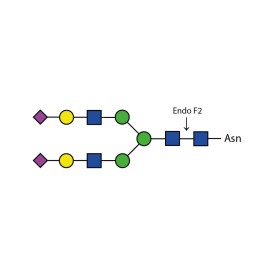
Endoglycosidase F2
E-EF02
LudgerLiberate Orela kit
LL-ORELA-A2
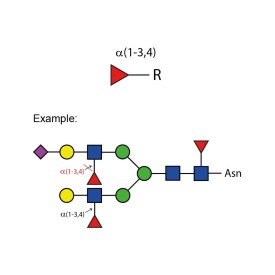
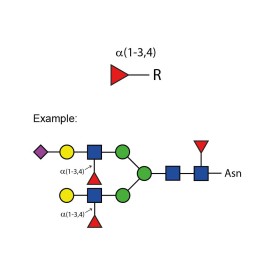
α-(1-3,4) fucosidase
E-F134
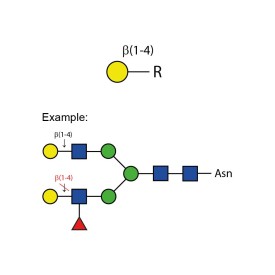
β-(1-4)-galactosidase
E-BG07
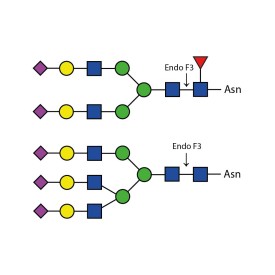
Endoglycosidase F3
E-EF03
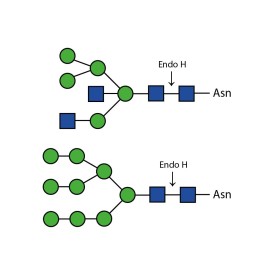
Endoglycosidase H
E-EH02
β-N-acetylglucosaminidase
E-GL01
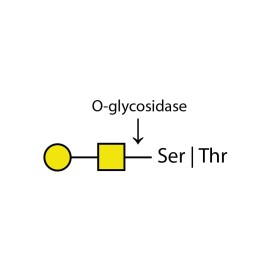
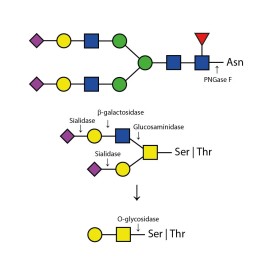
O-glycosidase
E-G001
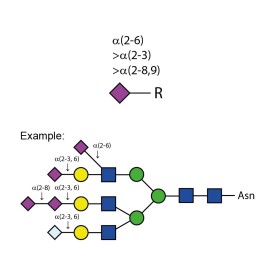
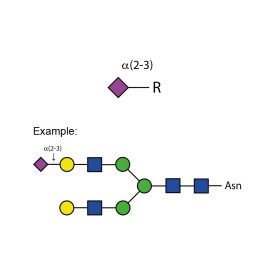
Sialidase Au α-(2-3,6,8,9)
E-S001
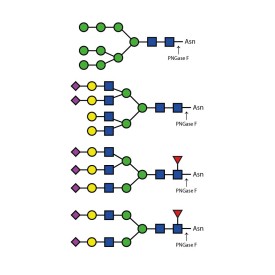
PNGase F (Peptide N Glycosidase F)
E-PNG01-200
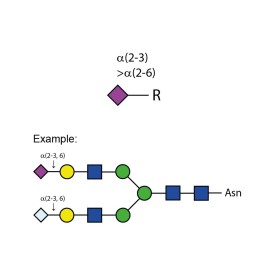
Sialidase Cp α-(2-3,6)
E-S005
Recombinant PNGase F
E-rPNG01
Sialidase Sp α-(2-3)
E-S007
Endo-β-galactosidase
E-XBG01
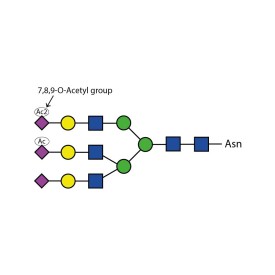
Sialate O-acetylesterase kit
LZ-ACASE-KIT
Enzymatic CarboRelease kit
KE-DG01
LudgerZyme α(1-3,4) fucosidase kit
LZ-FUCOSIDASE-01-KIT
Enzymatic DeGlycoMx Kit
KE-DGMX
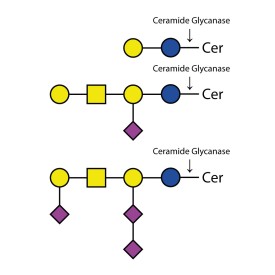
Ceramide Glycanase Kit
LZ-CER-HM-KIT
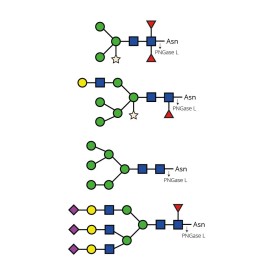
LudgerZyme PNGase L Kit
LZ-PNGaseL-50-KIT
Recombinant PNGase F
LZ-rPNGaseF-kit
Showing 1 to 26 of 26 (1 Pages)